Soap and detergent are the most common cleaning agent in our daily life. Soap and detergent provide convenience to us as they fasten our daily cleaning activity. They are substances that when dissolved in water, gained the ability to remove dirt from surfaces such as human skin, textiles and other solids.
Soap is a salt of a long-chain fatty acid which is produced from the hydrolysis of fats or oils under alkaline condition. The chemical reaction is called saponification. The main sources of fats are beef and mutton tallow, while the main sources of oils are palm, coconut, and also palm kernel oils. The raw materials are pretreated to remove impurities and to achieve the color, odor an performance feature desired in the finished soap. Saponification, in technical terms, involves the use of a base to hydrolyse triglyceride, which are esters of fatty acids to form the potassium or sodium salt of the carboxylate. Saponification also can be known as alkaline hydrolysis of ester. If the base used is sodium hydroxide , then sodium soap will be formed and it is in solid form. If the base used is potassium hydroxide, then potassium soap will be formed which will be smoother and is found in liquid form.
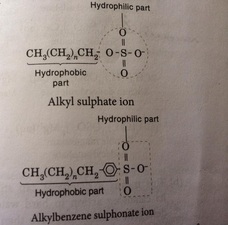
Any cleaning agent that is not a soap is a detergent. Detergent are usually made from synthetic resources such as petroleum fractions. Detergent were developed during the Second World War in response to a shortage of animal fats and vegetable oil. Detergent are usually sodium salts of sulphonic acid. During the preparation of detergents, long chain hydrocarbon obtained from petroleum fractions is converted into an organic acid through a series of steps. The organic acid then undergo neutralization by using sodium hydroxide solution to produce a neutral salt which is a detergent. Detergent usually exist in two form which is powder or liquid form.
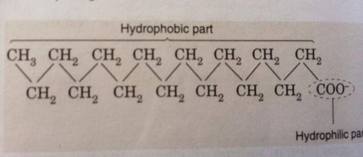
Soap and detergent are good cleaning agent as they can act as emulsifying agent. An emulsifier is a molecule that contains two different ends. One of the end is a hydrophilic head which is attracted to water, while another end is a hydrophobic tail which is repelled by water or ‘water hating’ and also ‘fat-loving’ . An emulsifier is capable of dispersing one liquid into another immiscible liquid. When water and oil are mixed together, they will usually separate into two distinct layers. Hence, an emulsifier is needed to forces the oil and water to mix at which make the oil droplets tend to stay in water for a longer time. When soap and detergent are added into water, they dissolved to form soap anions or detergent anions and sodium cations. Soap anion consists of a long hydrocarbon chain with a carboxylate group on one end and the hydrocarbon chain consist of a hydrophobic part and a hydrophilic part. Detergent anion also consists of a hydrophobic part and a hydrophilic part.
Soap is a salt of a long-chain fatty acid which is produced from the hydrolysis of fats or oils under alkaline condition. The chemical reaction is called saponification. The main sources of fats are beef and mutton tallow, while the main sources of oils are palm, coconut, and also palm kernel oils. The raw materials are pretreated to remove impurities and to achieve the color, odor an performance feature desired in the finished soap. Saponification, in technical terms, involves the use of a base to hydrolyse triglyceride, which are esters of fatty acids to form the potassium or sodium salt of the carboxylate. Saponification also can be known as alkaline hydrolysis of ester. If the base used is sodium hydroxide , then sodium soap will be formed and it is in solid form. If the base used is potassium hydroxide, then potassium soap will be formed which will be smoother and is found in liquid form.
Any cleaning agent that is not a soap is a detergent. Detergent are usually made from synthetic resources such as petroleum fractions. Detergent were developed during the Second World War in response to a shortage of animal fats and vegetable oil. Detergent are usually sodium salts of sulphonic acid. During the preparation of detergents, long chain hydrocarbon obtained from petroleum fractions is converted into an organic acid through a series of steps. The organic acid then undergo neutralization by using sodium hydroxide solution to produce a neutral salt which is a detergent. Detergent usually exist in two form which is powder or liquid form.
Soap and detergent are good cleaning agent as they can act as emulsifying agent. An emulsifier is a molecule that contains two different ends. One of the end is a hydrophilic head which is attracted to water, while another end is a hydrophobic tail which is repelled by water or ‘water hating’ and also ‘fat-loving’ . An emulsifier is capable of dispersing one liquid into another immiscible liquid. When water and oil are mixed together, they will usually separate into two distinct layers. Hence, an emulsifier is needed to forces the oil and water to mix at which make the oil droplets tend to stay in water for a longer time. When soap and detergent are added into water, they dissolved to form soap anions or detergent anions and sodium cations. Soap anion consists of a long hydrocarbon chain with a carboxylate group on one end and the hydrocarbon chain consist of a hydrophobic part and a hydrophilic part. Detergent anion also consists of a hydrophobic part and a hydrophilic part.
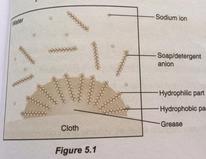
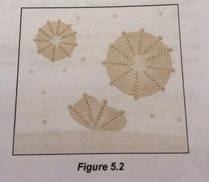
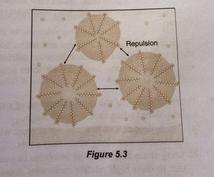
When soap or detergent are added into the water, they lowered the surface tension of the water. The hydrophobic part of soap or detergent is soluble in grease while the hydrophilic part is soluble in water. Mechanical agitation during scrubbing helps to pull the grease free and also break the grease into small droplets. The droplets do not coagulate and redeposit on the surface of the solids due to repulsion between negative charges on their surface. These droplets are suspended in water, forming an emulsion and rinsing washes away those droplets and leave the surface clean.
The cleansing action of detergent is more effective than a soap. Although soap is a good cleaning agent, its effectiveness reduced when the water used is hard water and acidic water. Hard water is type of water which contain great amount of calcium and magnesium ions. These ions will react with the soap to form insoluble precipitate known as soap scum. Formation of those soap scum reduced the amount of soap available for cleaning. When acidic water is used, the hydrogen ion in acidic water react with soap to form long-chain fatty acids which is insoluble due to their high relative molecular masses. Hence, soap is only effective in soft water. Detergents do not form scum with hard water and form precipitate in acidic water. Detergent will form soluble substances with calcium and magnesium ion in hard water and will form soluble precipitate in acidic water. Thus, the cleansing action of detergent is not affected and is more effective than soap.