Introduction of Alcohol
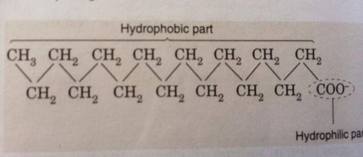
Alcohol actually is a organic compound contain hydroxyl group(OH) bond with carbon in chemistry .The general formula of alcohol is CnH2n+1 OH .In general, the hydroxyl group makes alcohol molecule polar. The hydrogen bond in the alcohol means it can soluble into the water . However, there are two opposing solubility in alcohol which the hydroxyl group tend to soluble in water and carbon chain resist to soluble in water. Thus , methanol, ethanol and propanol are miscible in water . Butanol is moderately soluble in water as the trends in this molecule is same . The alcohol contain five and more carbon are insoluble in water as the carbon chain has became the dominance in the molecule. The alcohol have higher boiling point than hydrocarbon because it contain hydrogen bonds. The boiling point of ethanol is 78.29 °C ,while the boiling point of hexane just 69 °C.
Alcohol actually is a organic compound contain hydroxyl group(OH) bond with carbon in chemistry .The general formula of alcohol is CnH2n+1 OH .In general, the hydroxyl group makes alcohol molecule polar. The hydrogen bond in the alcohol means it can soluble into the water . However, there are two opposing solubility in alcohol which the hydroxyl group tend to soluble in water and carbon chain resist to soluble in water. Thus , methanol, ethanol and propanol are miscible in water . Butanol is moderately soluble in water as the trends in this molecule is same . The alcohol contain five and more carbon are insoluble in water as the carbon chain has became the dominance in the molecule. The alcohol have higher boiling point than hydrocarbon because it contain hydrogen bonds. The boiling point of ethanol is 78.29 °C ,while the boiling point of hexane just 69 °C.
History of alcohol
From past until today, alcohol is a product that has provided a variety of functions for people throughout all history. Historically, alcoholic beverages have served as sources of needed nutrients and have been widely used for medical, antiseptic, analgesic and most importantly it has become one of the most important drinks in the world. The word “alcohol” appears in English as a term for a very fine powder in the 16th century. It was borrowed from French, which took it from medical Latin. In the early of Egyptian civilization, there is evidence of an early alcoholic drinks in China around 7000 B.C. On the other hand, in India , an alcoholic beverage called sura, distilled from rice, was in use between 3000 and 2000 B.C.
From past until today, alcohol is a product that has provided a variety of functions for people throughout all history. Historically, alcoholic beverages have served as sources of needed nutrients and have been widely used for medical, antiseptic, analgesic and most importantly it has become one of the most important drinks in the world. The word “alcohol” appears in English as a term for a very fine powder in the 16th century. It was borrowed from French, which took it from medical Latin. In the early of Egyptian civilization, there is evidence of an early alcoholic drinks in China around 7000 B.C. On the other hand, in India , an alcoholic beverage called sura, distilled from rice, was in use between 3000 and 2000 B.C.
Preparation of alcohol
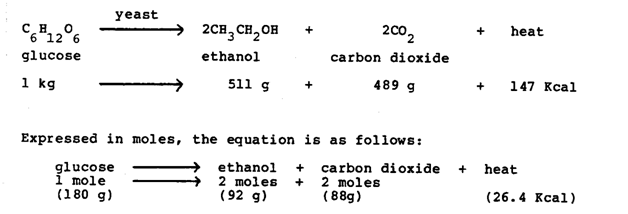
The preparation of alcohol can undergo hydration of alkenes, reduction of Aldehydes and Ketones and reduction of carboxylic acid .Also, there are a process called fermentation in biological routes . Many other alcohols can be made this way, but are more likely to be produced by synthetic routes - from natural gas, oil or coal. When yeast ferments, it breaks down the glucose (C6H12O6) into exactly two molecules of ethanol (CH3CH2OH) and two molecules ofcarbon dioxide (CO2).
The preparation of alcohol can undergo hydration of alkenes, reduction of Aldehydes and Ketones and reduction of carboxylic acid .Also, there are a process called fermentation in biological routes . Many other alcohols can be made this way, but are more likely to be produced by synthetic routes - from natural gas, oil or coal. When yeast ferments, it breaks down the glucose (C6H12O6) into exactly two molecules of ethanol (CH3CH2OH) and two molecules ofcarbon dioxide (CO2).
Ethanol fermentation (performed by yeast and some types of bacteria) breaks the pyruvate down into ethanol and carbon dioxide. It is important in bread-making, brewing, and wine-making. Usually only one of the products is desired; in bread-making, the alcohol is baked out, and, in alcohol production, the carbon dioxide is released into the atmosphere or used for carbonating the beverage. When the ferment has a high concentration of pectin, minute quantities of methanol can be produced. To make it simple, yeast eats sugar and makes alcohol and CO2.
Reaction of alcohol
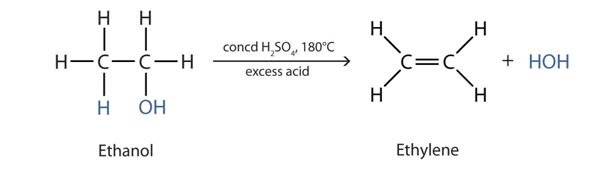
1) Alcohol can undergo dehydration reaction to give out alkene and water .
For example: Ethanol undergo dehydration to form ethylene and water .
1) Alcohol can undergo dehydration reaction to give out alkene and water .
For example: Ethanol undergo dehydration to form ethylene and water .
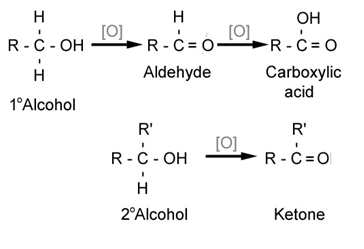
2) Alcohols can also undergo oxidation to give aldehydes, ketones, or carboxylic acids.
eg:
eg:
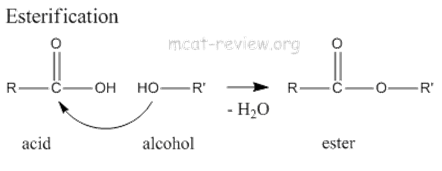
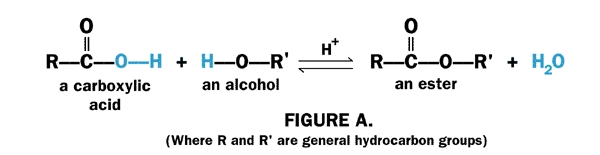
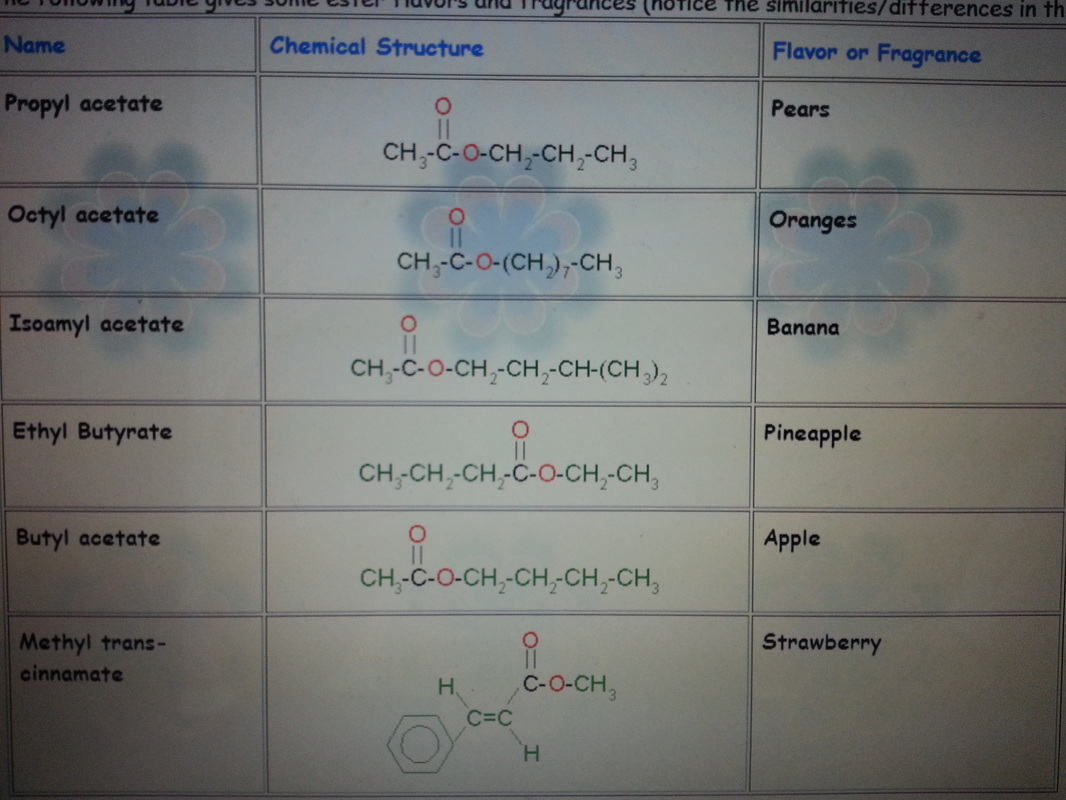
3) Alcohol and react with carboxylic acid to form ester through the Esterification reaction.
eg:
eg: